Wellbutrin vs. Generic Bupropion Review
Generic Bupropion Is Not
Always The Same As Name-Brand Wellbutrin. CL Tests
Reveal Differences Between Generic Bupropion and Name-Brand Wellbutrin.
Medically reviewed and
edited by Tod Cooperman, M.D. 
Last Updated![]() : 10/17/2013 | Initially Posted:
10/12/2007
: 10/17/2013 | Initially Posted:
10/12/2007

Recent Reviews
·
Aloe Juices, Gels, and Supplements
Review
·
NAD Booster Supplements Review
(NAD+/NADH, Nicotinamide Riboside, and NMN)
·
PQQ (Pyrroloquinoline Quinone)
Supplements Review
Table of Contents
Update
Update:
On October 10, 2013 the FDA updated its information about
bupropion hydrochloride ER 300 bioequivalence studies indicating that it
completed its review of the final two generic versions of this drug on the
market. The Actavis version was found to be bioequivalent. However, the Watson
version was found to not be therapeutically equivalent to Wellbutrin
XL 300 mg. Watson has agreed to voluntarily withdraw this product from the
market. The FDA noted that Watson recently merged with Actavis.
On August 22, 2013, Mylan Inc. announced
approval by the FDA of its version of bupropion hydrocholoride ER (XL) 300
based on a study it recently performed showing bioequivalence to Wellbutrin XL
300 mg. Par Pharmaceuticals made
a similar announcement on August 20 regarding its version of this drug
(originally produced by Anchen Pharmaceuticals, which
Par acquired). These product have been on the market
for several years but, as noted below, had originally been approved without
bioequivalence testing. These appear to be the first of the four generic
versions of Wellbutrin XL 300 on the market to be re-approved under a
"supplemental" application to the FDA (or "sANDA").
It should be noted that the FDA did not require the products to be tested when
taken with food, only on an empty stomach, to simplify comparisons between
drugs.
On December 5, 2012, the New England Journal of Medicine published a paper from
FDA officials entitled "Withdrawal of Generic Budeprion
for Nonbioequivalence-- reiterating
information from the FDA's October 3 announcement (below) but also revealing
that "certain study participants" in the FDA's recent bioequivalence
study achieved "less than 40% -- of the plasma levels of bupropion when
taking Teva's generic Budeprion XL 300 compared to
Wellbutrin XL 300. The number of participants affected this way is not given,
but even if just two of the 24 healthy individuals in the study, it suggests
that at least 8 percent of individuals taking the generic do not reach half the
amount of drug in their blood as with Wellbutrin XL. Considering the many
thousands of people switched to Budeprion XL 300, it
is now evident why so many reported dramatic changes in their mood.
On October 3, 2012 the FDA announced that Teva's Budeprion XL 300 is not bioequivalent to
Wellbutrin XL 300 mg. The reversal was based on a recent FDA
bioequivalence study which found Budeprion XL 300
achieved only 75% of the
maximum drug concentration attained with Wellbutrin XL 300 mg (potentially
explaining the return of depression in patients switched to Budeprion
XL 300). [Complete bioequivalence results as well as dissolution results --
which closely match those below from ConsumerLab.com -- were published by the FDA on 10/11/12.]
The FDA noted that Teva's voluntary bioequivalence study (discussed below) was
never completed. Teva and Impax have stopped shipping the product. The FDA is
requiring makers of the other four marketed generics — Anchen,
Actavis, Watson, and Mylan — to conduct bioequivalence studies, admitting that
each of these products had been approved on the basis of tests using 150 mg
versions. (See ConsumerLab.com's news release.)
On April 16, 2008 the FDA issued a news release affirming its position
that Teva's Budeprion XL 300 is safe and effective
based on its review. Analysis of the accompanying report,
however, revealed that the FDA did not conduct any tests of the product
and, in fact, does not have clinical studies on the product's safety,
efficacy, or bioequivalence. ConsumerLab.com issued a news release calling
the FDA's report incomplete and misleading.
From the FDA report, ConsumerLab.com determined that the some the information in the Budeprion
XL package insert is incorrect and misleading. For example, the Absorption section
of the insert (dated 6/2007) states that "Budeprion
XL" is not affected by food. However, the FDA report
suggests that, when taken with food, more drug is absorbed with Budeprion XL than with Wellbutrin XL. The Budeprion XL insert also refers to a 14-day
clinical study of Budeprion XL 300, while the
FDA report notes that no such study has been conducted. This and other
information in the Budeprion XL insert are apparently
based on data relating to brand-name Wellbutrin XL, not Budeprion
XL. In July 2008 ConsumerLab.com requested that the FDA take action to correct
this error. On October 2, 2008 it was learned from a Dow Jones article that the
FDA acknowledged labeling the inconsistencies but considers the product to be
"labeled appropriately." As explained by an FDA spokeswoman, the
labeling of a generic drug must match that of the drug it copies and this is
"the standard" for all generics. A week earlier, the FDA acknowledged
that it is considering new tests of Budeprion XL 300
to help understand reported problems (More information in Recalls & Warnings posting of October
2, 2008.) However, according to a December 2, 2009 Wall Street Journal article, the study
will not be conducted by the FDA but by Teva, and it will not investigate the
effect of taking Budeprion XL 300 with food. ConsumerLab.com
considers this a major omission considering the known food-effect noted in the
FDA's 2008 report on Budeprion XL 150 - which uses
the same pill formulation as the XL 300.
The Director of the Office of Medical Policy of the FDA's Center for Drug
Evaluation and Research, Dr. Robert Temple, has acknowledged that TEVA Budeprion XL 300 releases drug at a different rate than the
brand name Wellbutrin XL 300, consistent with ConsumerLab.com's
findings in this report. Dr. Temple's remarks were made during a radio broadcast on
Southern California Public Radio station KPCC on December 19, 2007. He noted
that the FDA was aware that bioequivalence studies in people showed the
once-a-day generic to release drug sooner than its brand-name counterpart but
that the FDA "thought that that wouldn't make any difference." He
admitted that the once-a-day generic "had an early release pattern that
was a little closer to the original [three-times-a-day] product" than to
once-a-day Wellbutrin XL. The FDA approved the once-a-day generic without
mention of this difference, further clinical testing or special follow up.
Two weeks prior to Dr. Temple's remarks, on December 1, 2007, the United States
Pharmacopeia (USP) officially released information indicating that the
dissolution of the Budeprion XL 300 varied
significantly from the original drug. According to the USP, tablets of
Wellbutrin XL release only a small amount (less than 20%) of drug during the
first two hours of dissolution. In contrast, the Budeprion
XL releases at least 25% and as much as 50% of its drug in that period. These
USP dissolution standards are based upon performance characteristics that the
FDA approved for the original and generic drugs, respectively, and were
provided to USP by the manufacturers.
The new information from the FDA and USP corroborate ConsumerLab.com's
findings for the Budeprion XL 300 product in this
report which show it to release four times as much drug as the original
Wellbutrin XL 300 — 34% versus 8%, respectively — in the first two hours of a
dissolution test. The Generic Pharmaceutical Association questioned ConsumerLab.com's findings. It is now clear that ConsumerLab.com's findings were consistent with differences
well known to both the manufacturer and the government but not disclosed
publicly.
Concerns about Generic
Wellbutrin:
Background:
Generic drugs can make prescription medication much more affordable. But in
early 2007, ConsumerLab.com became aware of reports by consumers experiencing
problems when switching from Wellbutrin to its generic form (bupropion HCl).
Wellbutrin is among the most popular anti-depressants. U.S. sales in 2006 were
in excess of $1.8 billion according to Wolters Kluwer Health. The majority of
these sales were of the XL (once-a-day) 300 mg product. Most consumer
complaints were about a generic version of the XL 300 mg product launched in
December 2006 as Budeprion XL 300 mg by Teva
Pharmaceutical Industries.
Consumer Complaints:
Early this year, The People's Pharmacy, a syndicated radio
program and newspaper column, began collecting and reporting consumer
complaints about generic Wellbutrin, many of which can be viewed on its website
at www.peoplespharmacy.com. The personal
accounts generally indicated that while taking the brand name Wellbutrin XL 300
for months or years, people felt well and their psychological symptoms of
depression were successfully controlled. After switching to the generic
formulation, Budeprion XL 300, many reported symptoms
such as headaches, irritability, nausea and insomnia — known side-effects of
bupropion. Others shared stories of becoming easily upset or aggressive,
crying, gaining weight or experiencing a return of depressive symptoms. Some
reported thoughts of suicide while taking the generic form of Wellbutrin. A
large number of accounts note that returning to the original product brought
symptoms under control. These reports prompted ConsumerLab.com to investigate
whether extended-release (XL) and sustained-released (SR) bupropion generics
were truly equivalent to original Wellbutrin and to each other.
Different Pills — But Called Equivalent:
The active ingredient in Wellbutrin is off-patent while the method for
delivering it in time-released tablets is still patent protected. As a result,
companies wishing to market their own time-released bupropion generics have had
to use or develop types of tablets different from those sold as Wellbutrin XL
or SR or obtain a license from the originator. In fact, as of September 2007,
many of the time-released generics of Wellbutrin on the market used different
tablet technologies.
Different types of tablets can release active ingredient at different rates.
This can affect a drug's performance. But the safety and efficacy of generic
drugs, unlike original drugs, is not clinically tested. Instead, the FDA relies
on data from each manufacturer showing that their generic contains the same
ingredient as the original, dissolves at approximately the same rate, and
appears in people's blood at about the same levels as with the original
product. If the results are within the FDA's limits, the generic is deemed
"bioequivalent" to the original drug and can be approved for sale.
However, reviewing the FDA's test requirements, ConsumerLab.com found that
generic products are allowed to vary from one another by a fairly wide range.
For example, a twice-a-day (SR) version of bupropion is permitted to release
anywhere from 60% to 85% of its ingredient after four hours into a dissolution
test. At the completion of such a test (which only runs eight hours), a product
could be as little as 80% dissolved or over 100% dissolved and still be considered
bioequivalent. ConsumerLab also discovered that the
acceptable limits for once-a-day (XL) versions have been blacked-out or deleted
in documents on the FDA website.
For a list of other popular time-released generic drugs, see Extended Release
Generics — Are They the Same as the Original?
Potential for Side-Effects:
A particular concern with Wellbutrin has been the potential for side-effects,
including seizures if a high dose is provided too quickly. For this reason,
instructions for Wellbutrin SR 150 mg (twice-a-day) state that doses should be
given at least eight hours apart. It is, therefore, important that any version
of bupropion release ingredient slowly enough to avoid delivering a large
amount too quickly.
How the Products Were
Tested:
ConsumerLab.com
purchased the original Wellbutrin XL 300 mg product sold by GlaxoSmithKline as
well as generic Budeprion XL 300 mg sold by Teva
Pharmaceuticals. ConsumerLab.com also purchased two generic bupropion SR
products. One, a SR 150 mg product from Watson Pharmaceuticals, is actually
supplied to Watson by GlaxoSmithKline, the maker of Wellbutrin SR, and, aside
from markings on the pills, is identical to 150 mg Wellbutrin SR. The other SR
product was a 200 mg SR bupropion distributed by Global Pharmaceuticals that
uses a different tablet technology. All products were first tested to establish
that they contained the correct amount of bupropion hydrochloride. The tablets
then underwent dissolution testing — to determine the rates at which the
tablets released active ingredient over specific periods of time. Dissolution
testing of the XL products followed the method specified by the FDA in its
approval of both Wellbutrin XL and the generic version sold by Teva. The two SR
products were tested using a method developed by the United States Pharmacopeia
(USP) specifically for SR products. See How Products Were Evaluated for more
information about the testing.
What CL Found:
Analyses of all four products showed that each contained its
claimed amount of bupropion hydrochloride. However, the rates at which the
products released their ingredient were quite different, raising concerns about
their equivalency. A second laboratory repeated the dissolution tests on all of
the products. Results from the second lab confirmed the major differences found
in the first lab which are described below. XL (Once-a-Day)
Versions:
·
Wellbutrin XL 300 mg
·
Teva Budeprion XL 300 mg (Generic
Bupropion)
|
|
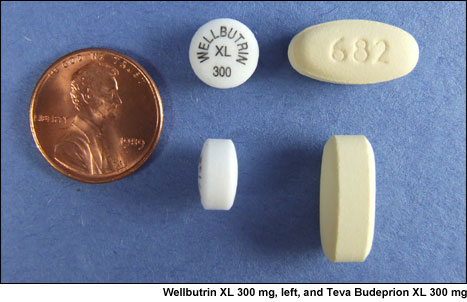
Even before testing began, it was clear that these two products were not alike.
Teva's XL 300 mg is a much larger pill than Wellbutrin XL 300 mg and found to
weigh more than twice as much. These differences are due to the fact that these
products use very different drug delivery technology. The Teva pill, made by
IMPAX Laboratories, uses a matrix of materials that expands when wet and then
breaks apart to release the ingredient. Wellbutrin XL, made by Biovail Corporation for GlaxoSmithKline, consists of a membrane
through which the ingredient is slowly released while the membrane stays
intact.
|
|
As seen in the graph above, the Teva product released much of its drug earlier
than Wellbutrin. Wellbutrin (shown in blue) released ingredient into solution
slowly over the first several hours of the dissolution test, with only 8%
dissolved at two hours and 25% at four hours. In contrast, Teva's pill released
ingredient rapidly at first — delivering over four times as much drug as
Wellbutrin in the first two hours (34% vs. 8%). Within the first four hours,
nearly half the drug in the Teva product had gone into solution, almost twice
as much as Wellbutrin. At eight hours into the test, results for Teva and
Wellbutrin began to even out, with each about three-quarters dissolved. By the
end of the test, both products had fully released their expected amounts of drug.
As noted earlier, the FDA has hidden from online viewing the specific
dissolution requirements for generic forms of Wellbutrin XL. So
it's not clear if the above results for Teva meet the FDA's requirements for bioequivalency.
What This Could Mean to You:
Consumer complaints about Teva Budeprion XL 300 mg
suggest that, at least for some people, it acts differently than the original.
And it is clear that the two products behaved differently in the dissolution
test. Do the test differences explain the reported problems? It's difficult to
know. By dissolving more rapidly than Wellbutrin within the first few hours, it
is possible that blood levels of the generic drug might at first rise higher
than with Wellbutrin, potentially increasing the risk of side-effects. As much
of this early burst of drug is eliminated over the rest of the day, the amount
of drug in the blood might then fall lower than with Wellbutrin, potentially
making the Teva product relatively less effective later on.
SR (Twice-a-Day) Versions:
·
Global Generic Bupropion SR 200 mg
·
Watson Generic Bupropion SR 150 mg
|
|
These two SR (twice-a-day) products were compared because, even though their
strengths are slightly different (200 mg vs. 150 mg), they should release their
ingredients at approximately the same rate. However, like the XL products,
these two SR (twice-a-day) products are made with different tablet technologies
from different companies. The Watson generic product is actually identical to
Wellbutrin SR 150 mg. It is supplied by GlaxoSmithKline to Watson to sell as an
"authorized generic." It is made with tablet technology from Biovail Corporation, like Wellbutrin SR. Global
Pharmaceutical's SR 200 mg, however, is made with different technology and is
manufactured by IMPAX Laboratories. (IMPAX is the same company that makes
Teva's 300 mg XL product described above.)
|
|
As shown in the graph above, the Global SR 200 mg yielded a smaller percentage
of its claimed amount of bupropion than the Watson SR 150 mg product at every
point in time. In fact, at two hours, Watson released 65% of its ingredient,
while Global released only 45%. At four and six hours, Watson also released
more of its ingredient — eighteen percentage points more. At eight hours,
Watson had released 102% of its expected ingredient versus only 90% for
Global.
The test results for both SR products fall within the FDA's acceptable range
established by the United States Pharmacopeia (USP) (see How Products Were Evaluated for more
information). However, Watson was near the upper limits for dissolution while
Global was close to the lower limits. The Watson SR provided more of its total
ingredient and released it faster.
What This Could Mean to You:
To date, ConsumerLab.com is not aware of consumer complaints about the Global
SR product. But differences between it and the Watson product should be kept in
mind.
Watson's bupropion SR products (which include the 150 mg tested, as well as 100
mg and 200 mg tablets) are supplied to it by GlaxoSmithKline and should,
therefore, be identical to the corresponding Wellbutrin SR products. These SR
products are all made with technology from Biovail
Corporation and it is likely that they dissolve in a manner similar to one
another.
Since the Global SR 200 mg product released less of its active ingredient than
the Watson product during the eight hour dissolution
test, it is possible that the Global SR 200 mg may also provide somewhat less
drug if tested against Watson's SR 200 mg or Wellbutrin SR 200 mg, potentially
making it less potent early on. It is also possible, however, that the Global
product eventually releases the same amount of ingredient as these drugs, just
taking a little longer, which could actually sustain its effectiveness later in
the day.
Summary of
Wellbutrin/Bupropion Tablet Dissolution Tests:
|
Type
and Strength |
Different
from Original? |
Test
Results |
What
This Could Mean to You! |
|
|
XL (Once-a-Day): |
||||
|
Wellbutrin |
XL 300 mg |
Original. |
Releases slowly during first few hours; more rapidly
between 4 and 8 hours; slowing thereafter. |
The original product. |
|
Budeprion |
XL 300 mg |
Different type of pill and delivery technology. Pill is
three times heavier than original. |
Releases rapidly in first few hours, but less over rest of
time period. |
Rapid initial release of drug may increase risk of
side-effects; a lower level later in day might provide a less effective level
of drug. Consumer complaints reported. |
|
SR (Twice-a-Day): |
||||
|
Bupropion |
SR 200 mg |
Different type of pill and delivery technology. |
Releases slightly lower percentage of its drug than other
SR product during eight-hour test. |
Meets FDA requirement. May release slightly less
ingredient initially, but no problems known to be reported. |
|
Bupropion |
SR 150 mg |
Same as Wellbutrin 150 SR— but sold as an "authorized
generic." |
Releases full amount of ingredient over time. |
Same as original product. Releases full amount of
ingredient. |
|
Note: |
||||
A Look at Other FDA
Approved Bupropion Generics:
Additional bupropion products have been approved by the FDA and
many are on the market. However, they are not necessarily identical to the
comparable version of Wellbutrin — often differing in their tablet technology
(i.e., the way in which drug is released). Be aware that the generic market is
fast-changing: Products and their availability may change. Brands carried will
also vary by pharmacy. Other Generic Buproprion
Hydrochloride Products (Not Tested by ConsumerLab.com)
|
Drug and Distributor |
Type
and Strength |
Different
from Original? |
What
This Could Mean to You! |
|
XL (Once-a-Day): |
|||
|
Bupropion |
XL 300 mg |
Different type of pill and delivery technology. |
Not identical to Wellbutrin or other generic versions.
Some difference in performance possible. |
|
Bupropion |
XL 300 mg |
Different type of pill and delivery technology |
Not identical to Wellbutrin or other generic versions.
Some difference in performance possible. |
|
Budeprion |
XL 150 mg |
Different type of pill and delivery technology |
Not identical to Wellbutrin or other generic versions.
Some difference in performance possible. |
|
Budeprion |
XL 150 mg |
Different type of pill and delivery technology |
If similar to Teva's 300 mg tested in this report, may
release differently than original. |
|
SR (Twice-a-Day): |
|||
|
Bupropion |
200 mg SR |
Different type of pill and delivery technology. |
Not identical to Wellbutrin or other generic versions.
Some difference in performance possible. |
|
Bupropion |
200 mg SR |
Same as Wellbutrin 200 SR — but sold as an
"authorized generic." |
Same as original product. |
|
Bupropion |
150 mg SR |
Different type of pill and delivery technology. |
Not identical to Wellbutrin or other generic versions.
Some difference in performance possible. |
|
Bupropion |
150 mg SR |
Same as Wellbutrin 150 SR — but sold as an
"authorized generic." |
Same as original product. |
|
Bupropion |
100 mg SR |
Different type of pill and delivery technology. |
Not identical to Wellbutrin or other generic versions.
Some difference in performance possible. |
|
Budeprion |
100 mg SR |
Different type of pill and delivery technology. |
Not identical to Wellbutrin or other generic versions.
Some difference in performance possible. |
|
Bupropion |
100 mg SR |
Same as Wellbutrin 100 SR— but sold as an "authorized
generic." |
Same as original product. |
|
Immediate release tablets: The original 3 times-a-day version of generic
bupropion is available in 75 mg and 100 mg strengths. The FDA has approved
such products from Teva, Mylan, Sandoz and Apotex.
Because immediate release tablets do not normally involve specialized drug
delivery technololgy, these products are likely to
behave similarly — although pills may vary in design. |
|||
Strategies for Getting
the Right Drug:
Request Generics by Name: If your current generic version is
working but others have not, make note of the distributor and the
manufacturer on the label and try to stick with that exact product. Ask your
pharmacist to fill your prescriptions with that specific version. Call around
if you need to — different pharmacies often carry different generic brands. Be
aware though that occasionally a generic brand may change its product due to
new licensing or manufacturing arrangements.
If you find that a generic version of Wellbutrin is not working, consider
the following:
·
Dispense as Written: Have your doctor prescribe the brand-name
version and indicate "Dispense as Written" or "Do Not
Substitute." If your insurance will not cover it, ask your doctor to make
a special request from your insurance company.
·
If your insurance still won't cover the original, you have other
options:
o Try a Different Generic: See if there is a
different generic equivalent — preferably one made by the same company that
makes the original drug, as this is likely to be identical to the original.
(See Other Generic Bupropion Products above)
o Get the Original at Low
Cost: Pay
out of pocket for the original, but try to save money by purchasing the drug
from a discount pharmacy or a licensed Canadian or other non-US pharmacy. You
can often save 50% or more over U.S. retail pharmacy prices. A three-month
supply (90 tablets) of Wellbutrin XL 300 mg, for example, costs about $550 from
a U.S. chain drug store but can be found for under $125 outside the U.S. — a
78% savings. A good place to compare low prices from licensed pharmacies
is www.PharmacyChecker.com, a site started by
ConsumerLab.com in 2002.
·
Switch Versions: Consider a different version of the
drug. If a once-a-day generic is not working, consider a twice-a-day (SR) or
three-times-a-day version that will yield the same total daily dose. You will
have to remember to take the medication on schedule.
ConsumerLab.com may test
additional versions of bupropion in the future. Stay up-to-date by checking
back or reading ConsumerLab.com's free e-newsletter,
which you receive as a subscriber.
If you have had a problem when switched to a generic drug, please let us
know. Click here.
Latest Clinical Research Updates for Wellbutrin vs. Generic
Bupropion
FDA Finds Another Generic Drug Not Equivalent
10/17/2013
According to the FDA, a
marketed generic version of the antidepressant buproprion
hydrochloride XL (300 mg) is not equivalent to the brand name
Wellbutrin XL and the product is being removed from the market. One year ago,
another generic version of this drug was removed from the market for the same
reason. ConsumerLab.com first reported potential problems with these products
in 2007 but the FDA was slow to take action. For details, see the updated in
the Wellbutrin vs Generic Budeprion Review >>
Failure of Generic Antidepressant Drug
12/09/2012
In October, the FDA
withdrew its approval of a generic version of the popular antidepressant drug,
Wellbutrin XL. This came 5 years after ConsumerLab.com first reported a
potential problem with the drug. This week, more disturbing details emerged
about this generic drug and why it was originally approved by the FDA. For more
about this, see the Update in the report Wellbutrin vs. Generic
Bupropion. More >>